NMDA Receptors
NMDA Receptors
What is the NMDA receptor?
Humankind is only at the dawn of understanding brain chemistry. The world’s most advanced computer lacks the creativity of a human brain. In part, that’s because electronic computers are completely binary in their logic circuits. Circuits are hard-wired to one another. Electrical connections are either on or off. The brain, however, is an analog computer in which both chemical and electrical signals modulate brain cells, which are constantly forming and breaking connections to one another.
The NMDA receptor is one example of a chemical pathway that modulates brain activity. Much like the volume control knob on an old-fashioned stereo, by controlling the rate at which Calcium ions do or do not enter the brain cell, the NMDA receptor. It is critical to memory function, as it controls signaling from neuron to neuron. NMDA also controls the rate of ideation (formation of new ideas) in the brain.
At excessive rates of NMDA activity, thoughts and the rate at which new thoughts are generated (ideation) is slowed. People tend to ruminate on those few thoughts, which are often negative and self-destructive in nature. When the NMDA channel is blocked, thoughts are generated rapidly and, often incoherently, leading to hallucinations and psychosis. The challenge is to strike a healthy balance and to maintain thought generation at a pace that is consistent with clear and creative thinking.
Much of the above was considered purely theoretical, based on observation of human and animal behavior but without direct biologic correlation until 2019 when Professor Connor Liston published a groundbreaking paper in Science, demonstrating the atrophy of dendritic spines on brain cells in the presence of high levels of NMDA activity. Dendritic spines are the “fingers” that brain cells use to touch other brain cells. Liston demonstrated that when ketamine, an NMDA antagonist drug was added to the experiment, the dendritic spines immediately regenerated and began forming new cellular connections. Scientists around the world have subsequently demonstrated that modulation of the NMDA receptor is key to the function and connectivity of thought-forming cells in the brain.
NMDA antagonists are potent antidepressants
In 2000, Professor Robert Berman at Yale University made the unexpected discovery that ketamine had potent antidepressant effects. He was conducting a human study in which ketamine was being administered to human volunteers to study its psychedelic effects. Unexpectedly, those volunteers reported that symptoms of depression were alleviated. This discovery provided context for the 1959 discovery by Professor George Crane that D-cycloserine, a newly-developed treatment for tuberculosis was a potent antidepressant. Crane was treating tuberculosis patients when he made the unexpected observation. He followed this observation with a placebo-controlled trial that demonstrated the antidepressant effect of D-cycloserine. At the time, however, there was no understanding of brain chemistry and NMDA receptors, much less the understanding that D-cycloserine is a potent NMDA receptor.
Anti-depressant research with D-cycloserine did not continue because another tuberculosis drug – Isoniazid – was shown to have antidepressant effects with no psychedelic effects and was developed into the first class of antidepressant drugs based on Iproniazid. These drugs, despite considerable toxicities, were discovered to raise serotonin. After the effect of NMDA inhibition was discovered, there was a resurgence of interest in D-cycloserine and other overlooked NMDA-antagonist medications, such as dextromethorphan and d-methadone. A comprehensive list of such medicines is elucidated in Javitt’s 2013 patent filings.
NMDA antagonists are shown to block suicidal thoughts
Recent studies demonstrate that NMDA antagonists – such as ketamine – directly inhibit suicidal thoughts, an effect that is distinct from their antidepressant effect.1,2,3 Hence, suicidal ideation though it may be present in psychiatric disorders such as bipolar depression, PTSD, and major depressive disorder, may be thought of as a distinct target (medical indication). The effect of ketamine on suicidal ideation was first shown by Zarate and coworkers at the U.S. National Institutes of Health and demonstrated in a large controlled trial by Professors Michael Grunebaum and Alfred Mann (a member of the NRx advisory board) at Columbia University.
The cost of studying ketamine in association with suicidal ideation is considerable, in that patients with acute suicidal ideation are frequently admitted to psychiatric hospitals. The French government funded the first large, multicenter trial of ketamine in patients with acute suicidal ideation at eight French hospitals and the results were reported by Professor Mocrane Abbar (a member of the NRx advisory board) together with his co-investigators. The trial documented both the expected effect of ketamine in reducing suicidal thoughts and the surprising discovery that the ketamine effect on suicidality was five times more potent among those with bipolar depression than among those with major depressive disorder.
Overcoming the psychedelic effects of NMDA drugs
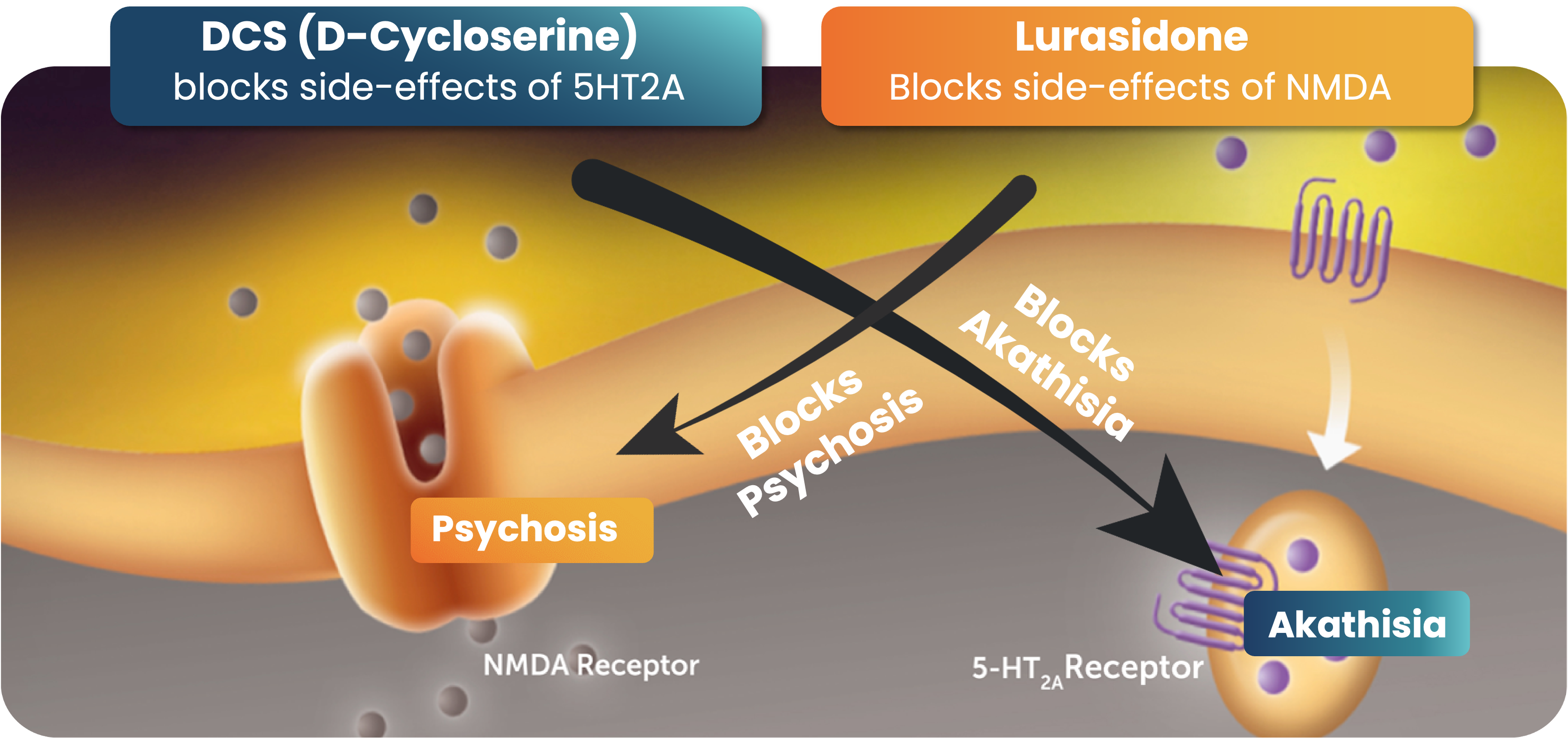
Although there has been interest in psychedelic therapy as a path to treating depression, PTSD, and suicidality, psychedelic effects are incompatible with a medicine for home use. Based on laboratory research that followed the first successful human trial with D-cycloserine for depression, Javitt discovered that the psychedelic effects of NMDA drugs can be reversed by the combination with serotonin-targeted drugs. He made the simultaneous discovery that NMDA inhibitors, in turn, block the akathisia side effect seen with all serotonin-targeted drugs. This key discovery has led to patents now-issued worldwide documenting the synergistic effect of NMDA antagonist and serotonin-targeting drugs.
- Malik, F. et al. Curr Psychiatry Rep, 2016: 18: 61
- Diaz-Granados, N. J Clin Psychiatry. 2010 Dec; 71(12): 1605–1611.
- Ballard, E. Journal of Psychiatric Research, 2014: 58:161-166